
The diagram below shows the arrangement of the atoms in each layer, and the way the layers are spaced. Diamond cutters take advantage of lines of cleavage to facet gemstones. Graphite has a layer structure which is quite difficult to draw convincingly in three dimensions. The lines of cleavage result from the diamond crystal having fewer chemical bonds along the plane of its octahedral face than in other directions. Diamonds have perfect cleavage in four different directions, meaning a diamond will separate neatly along these directions rather than break in a jagged manner. Real diamond crystals don't have completely smooth faces but may have raised or indented triangular growths called 'trigons'. 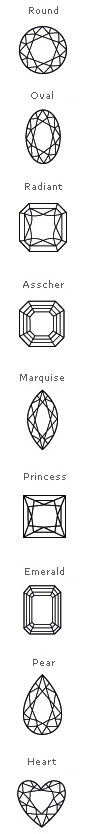
Diamond crystals can also form cubes, dodecahedra, and combinations of these shapes. Marquise, oval, and pear-shaped diamonds are more distinctive shapes, and help. The most common crystal habit is the eight-sided octahedron or diamond shape. They emit less fire or sparkle than round diamonds but are more transparent. One exception is the flat form called a macle, which is really a composite crystal, and the other exception is the class of etched crystals, which have rounded surfaces and may have elongated shapes. Based on the cubic form and its highly symmetrical arrangement of atoms, diamond crystals can develop into several different shapes, known as 'crystal habits'. The total Weight of This Black Diamond is 1 Carat. Going beyond the Type I and Type II classification, there are a few notable consumer-facing diamond types available on the market that don’t require a degree. Except for two shape classes, these structures are manifestations of the cubic crystal system. Hexagon Shape Black Diamond is Famous for its Geometrical Shapes. Additionally, there are fancy-shaped diamonds, including the princess cut. Diamond crystals can also form cubes, dodecahedra, and combinations of these shapes. However, the round brilliant-cut diamond is the most popular and traditional shape. The most common crystal habit is the eight-sided octahedron or diamond shape. Based on the cubic form and its highly symmetrical arrangement of atoms, diamond crystals can develop into several different shapes, known as 'crystal habits'. Each carbon atom joins four other carbon atoms in regular tetrahedrons (triangular prisms). There is a range of traditional and unique diamond ring shapes to truly. The crystal structure of a diamond is a face-centered cubic or FCC lattice. Diamond shape, often confused with diamond cut, refers to its geometric appearance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
